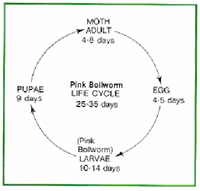
The pink bollworm has been eradicated from the southwestern United States (Figs. 74–77). Contact your local AgriLife Extension office for information.

Cotton boll heavily infested with pink bollworms; one or more boll locules may be damaged depending on the number of pink bollworm larvae or the incidence of boll rotting organisms